Familial Partial Lipodystrophy
Familial partial lipodystrophy – Dunnigan Variety is a rare genetic disease. It is transmitted as an autosomal dominant trait. Both males and females of several generations may be affected. The possibility of transmission from an affected person to the offspring is 50 %.
There is another variety of FPLD called Kobberling variety. In this Loss of fat is limited to the extremities with normal amounts of fat in the face and normal or even excess fat in the truncal area.
Clinical features
- Normal appearance at birth.
- Absence of subcutaneous fat from the upper and lower extremities during childhood or puberty. The trunk, arms and legs are affected by fat loss while the neck and face have more than normal fat deposits. A “buffalo hump” can be observed between the shoulder blades.
- The arms and legs appear very muscular.
- In women, lack of fat in the buttocks is striking — flat hips.
- In some patients, excess fat may accumulate in the face and neck, causing them to have a double chin.
- They usually have high levels of serum triglycerides and low HDL cholesterol levels.
- Approximately one-third of these patients may have acanthosis nigricans.
- The onset of glucose intolerance or diabetes mellitus usually occurs after age 20.
- Some women may have irregular menstrual cycles and polycystic ovaries.

Patients with Familial Partial Lipodystrophy of the Dunnigan Type. Source: http://jcem.endojournals.org/cgi/content/full/89/11/5337/F2
Acquired Generalized Lipodystrophy Congenital Generalized Lipodystrophy


 body can be affected although in some patients some areas may be spared.
body can be affected although in some patients some areas may be spared.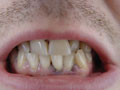 ability to concentrate.Encephalopathy is rare. A “lead line” sometimes appears at the gingiva-tooth border after prolonged high-level exposure.Chronic subclinical lead exposure is associated with interstitial nephritis, tubular damage (with tubular inclusion bodies), hyperuricemia (with an increased risk of gout), and a decline in glomerular filtration rate and chronic renal failure.An additional issue for both children and adults is whether lead that has accumulated in bone and lain dormant for years can pose a threat later in life, particularly at times of increased bone resorption such as pregnancy, lactation, and senile osteoporosis. Elevation of the bone lead level appears to be a risk factor for anemia and hypertension. Hyperthyroidism has been reported to cause lead toxicity in adults by mobilizing stores of bone lead acquired during childhood.
ability to concentrate.Encephalopathy is rare. A “lead line” sometimes appears at the gingiva-tooth border after prolonged high-level exposure.Chronic subclinical lead exposure is associated with interstitial nephritis, tubular damage (with tubular inclusion bodies), hyperuricemia (with an increased risk of gout), and a decline in glomerular filtration rate and chronic renal failure.An additional issue for both children and adults is whether lead that has accumulated in bone and lain dormant for years can pose a threat later in life, particularly at times of increased bone resorption such as pregnancy, lactation, and senile osteoporosis. Elevation of the bone lead level appears to be a risk factor for anemia and hypertension. Hyperthyroidism has been reported to cause lead toxicity in adults by mobilizing stores of bone lead acquired during childhood. 1.9 umol/L (40 ug/dL) is advised.Lead-associated anemia is usually normocytic and normochromicand may be accompanied by basophilic stippling. Lead-induced peripheral demyelination is reflected by prolonged nerve conduction time and subsequent paralysis, usually of the extensor muscles of the hands and feet –wrist and foot drop. An increased density at the metaphyseal plate of growing long bones (“lead lines”) can develop in children and
1.9 umol/L (40 ug/dL) is advised.Lead-associated anemia is usually normocytic and normochromicand may be accompanied by basophilic stippling. Lead-induced peripheral demyelination is reflected by prolonged nerve conduction time and subsequent paralysis, usually of the extensor muscles of the hands and feet –wrist and foot drop. An increased density at the metaphyseal plate of growing long bones (“lead lines”) can develop in children and resemble those seen in rickets. Children with high-level lead exposure sometimes develop Fanconi’s syndrome, pyuria, and azotemia.Adults chronically exposed to lead can develop elevated serum creatinine levels, decreased creatinine clearance rates, and chronic changes and intranuclear inclusion bodies (detected at renal biopsy).
resemble those seen in rickets. Children with high-level lead exposure sometimes develop Fanconi’s syndrome, pyuria, and azotemia.Adults chronically exposed to lead can develop elevated serum creatinine levels, decreased creatinine clearance rates, and chronic changes and intranuclear inclusion bodies (detected at renal biopsy). 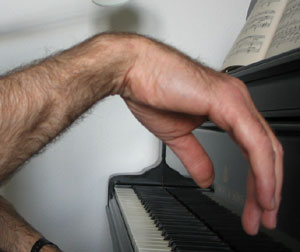
 inflammation and necrosis of the intestinal mucosa; these changes manifest as hemorrhagic gastroenteritis, fluid loss, and hypotension. Delayed cardiomyopathy accompanied by electrocardiographic abnormalities may develop. Symptoms include nausea, vomiting, diarrhea, abdominal pain, delirium, coma, and seizures. A garlicky odor may be detectable on the breath. Acute tubular necrosis and hemolysis may develop. The reported lethal dose of arsenic ranges from 120 to 200 mg in adults and is 2 mg/kg in children. Arsine gas causes severe hemolysis within 3 to 4 h of exposure and can lead to acute tubular necrosis and renal failure.In chronic arsenic poisoning, the onset of symptoms comes at 2 to 8 weeks. Typical findings are skin and nail changes, such as hyperkeratosis, hyperpigmentation, exfoliative dermatitis, and Mees’ lines (transverse white striae of the fingernails); sensory and motor polyneuritis manifesting as numbness and tingling in a “stocking-glove” distribution, distal weakness, and quadriplegia; and inflammation of the respiratory mucosa.Epidemiologic evidence has linked chronic
inflammation and necrosis of the intestinal mucosa; these changes manifest as hemorrhagic gastroenteritis, fluid loss, and hypotension. Delayed cardiomyopathy accompanied by electrocardiographic abnormalities may develop. Symptoms include nausea, vomiting, diarrhea, abdominal pain, delirium, coma, and seizures. A garlicky odor may be detectable on the breath. Acute tubular necrosis and hemolysis may develop. The reported lethal dose of arsenic ranges from 120 to 200 mg in adults and is 2 mg/kg in children. Arsine gas causes severe hemolysis within 3 to 4 h of exposure and can lead to acute tubular necrosis and renal failure.In chronic arsenic poisoning, the onset of symptoms comes at 2 to 8 weeks. Typical findings are skin and nail changes, such as hyperkeratosis, hyperpigmentation, exfoliative dermatitis, and Mees’ lines (transverse white striae of the fingernails); sensory and motor polyneuritis manifesting as numbness and tingling in a “stocking-glove” distribution, distal weakness, and quadriplegia; and inflammation of the respiratory mucosa.Epidemiologic evidence has linked chronic consumption of water containing arsenic at concentrations in the range of 10 to 1820 ppb with vasospasm and peripheral vascular insufficiency culminating in “blackfoot disease – a gangrenous condition affecting the extremities.Chronic arsenic exposure has also been associated with a greatly elevated risk of skin cancer and possibly of cancers of the lung, liver (angiosarcoma), bladder, kidney, and colon.
consumption of water containing arsenic at concentrations in the range of 10 to 1820 ppb with vasospasm and peripheral vascular insufficiency culminating in “blackfoot disease – a gangrenous condition affecting the extremities.Chronic arsenic exposure has also been associated with a greatly elevated risk of skin cancer and possibly of cancers of the lung, liver (angiosarcoma), bladder, kidney, and colon.
